How to Build a Beating Heart: Tissue Engineering Today
Twenty years ago the idea that a human heart could be grown from a patient’s own cells in a laboratory tank was science fiction. Today it is a clinical research field with several functional prototypes, multiple ongoing human trials in adjacent territories (heart valves, cardiac patches, vascularised muscle grafts), and a credible path — though not a short one — to whole-organ replacement therapy. The 2012 documentary How To Build A Beating Heart introduced general audiences to tissue engineering at a time when the field was still mostly inhabited by specialists. Geography Scout’s team has been tracking the science since, and Hugo Vasiliev — who did research-assistant work in a Boston tissue-engineering lab during his postgrad — led the rewatch.
The headline finding from our review is that the documentary aged better than most. Several of the techniques it foregrounded have moved from speculative demonstration to mainstream research and, in a couple of cases, to early-stage clinical use. Where it was wrong was almost always on the timeline — the science has moved more slowly than the show implied — but where it was right was on the underlying biology, which has held up.
The Premise: Growing What the Body Can’t Replace
The core insight that drives tissue engineering is that the human body is a perpetual self-repair system, but the repair toolkit is unevenly distributed. We rebuild liver, skin, blood and bone marrow continuously throughout life. We do not — without external help — rebuild heart muscle, neurons, or pancreatic islet cells in any clinically meaningful quantity. That asymmetry explains why heart attacks, spinal-cord injuries, and type-1 diabetes are all so consequential: the damaged tissue stays damaged.
Tissue engineering attempts to outsource the repair. The basic recipe is to take cells (often the patient’s own), provide them with a structural scaffold (sometimes synthetic, sometimes a decellularised donor organ), and supply the chemical and mechanical cues that prompt the cells to organise themselves into functional tissue. The recipe is conceptually simple. The execution remains one of the hardest problems in modern biomedicine.
The Decellularisation Breakthrough
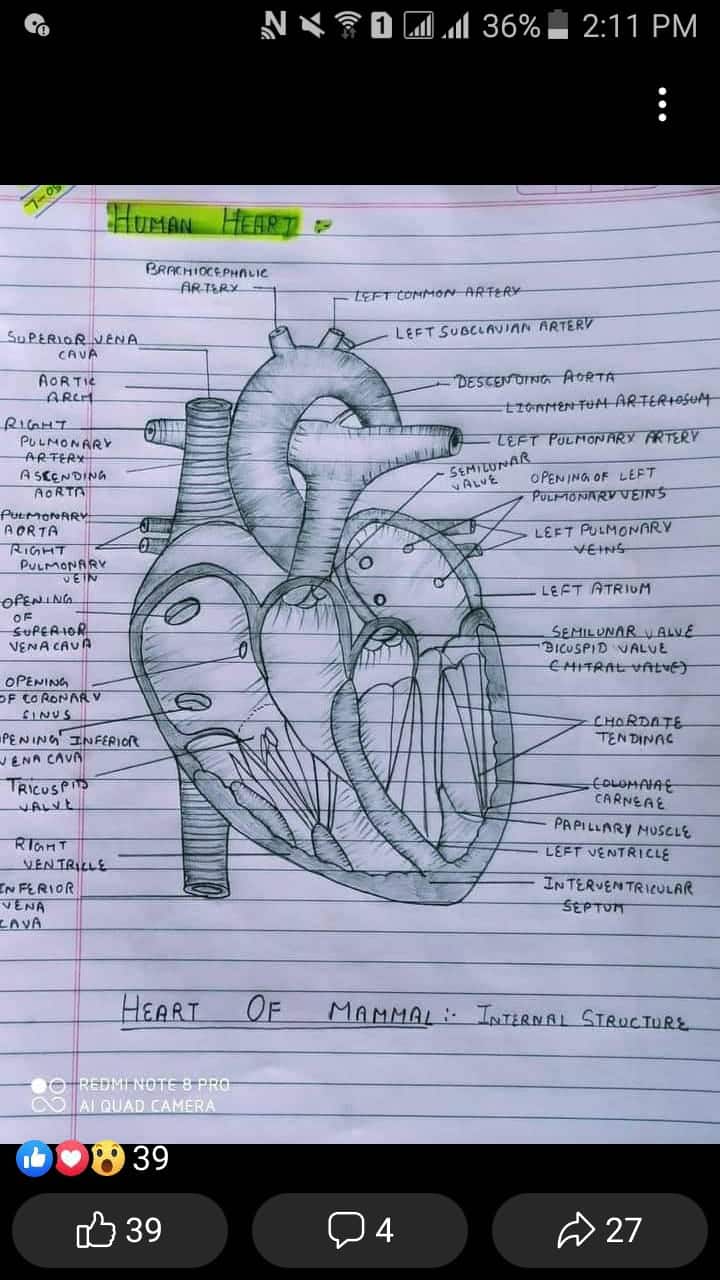
The single most important methodological innovation the documentary covers — and the one that has continued to drive the field since — is decellularisation. Pioneered by Doris Taylor and her team at the University of Minnesota in the late 2000s, the technique involves taking a donor organ (initially from a rat, since extended to pig and human cadaver hearts), and chemically washing away every cell while preserving the underlying extracellular matrix. What remains is a “ghost organ”: the structural scaffolding of a heart, complete with chambers, valves, vasculature, and connective tissue, but with none of the cells that would trigger immune rejection.
The ghost organ is then re-seeded with cells from the eventual recipient, ideally pluripotent stem cells differentiated into the multiple cell types a working heart requires (cardiomyocytes for muscle, endothelial cells for vessel linings, fibroblasts for connective tissue). The cells colonise the scaffold, organise themselves along the existing structural cues, and — in the most successful demonstrations — begin to beat. Taylor’s first decellularised-and-reseeded rat heart beat in vitro in 2008. The documentary covered her lab and showed the actual moment, which remains one of the better pieces of biomedical-research footage in the public domain.
The thirteen years since the broadcast have refined the technique substantially. Pig and human cadaver hearts have been successfully decellularised. The challenge that has slowed progress is reseeding at adequate density — a human heart contains roughly 2-3 billion cardiomyocytes and producing them in clinical quantities from stem cells is logistically demanding. Several groups (notably Harvard’s Wyss Institute and the Texas Heart Institute) are working on this problem with measurable progress.
Why the Heart Is Especially Hard
The heart is a difficult target organ for tissue engineering for three reasons that the documentary covers well. First, it has multiple cell types in close functional integration — you can’t grow heart muscle in isolation and then plumb it in afterwards. Second, the cells need to organise in a specific 3D pattern with directional alignment of the muscle fibres, which depends on both chemical cues (gradients of growth factors) and mechanical cues (cyclic stretching). Third, the heart is metabolically demanding — cardiomyocytes consume more oxygen per gram than almost any other tissue, which means the scaffold needs working vasculature from the moment the cells go in.
That last point is what has kept whole-heart tissue engineering in the laboratory rather than the operating theatre. Without functional capillary networks throughout the scaffold, even successfully seeded cells starve and die within days. The current research frontier is on creating those networks during the seeding phase — using endothelial cells co-seeded with the cardiomyocytes, or embedding micro-channels through 3D bioprinting techniques, or pre-vascularising the scaffold by surgical implantation in the recipient’s body for a maturation period before final reseeding.
The Adjacent Wins: Patches, Valves, and Vessels
While whole-heart engineering has progressed slowly, the adjacent technologies have moved faster, and that’s where the clinical impact has happened. Bioengineered heart valves have been in clinical use for over a decade. Decellularised allograft valves (most commonly from human donors, washed of cells and reseeded with the recipient’s own) are now standard care for several specific paediatric heart conditions, with documented superiority over older mechanical valves for younger patients (because the bioengineered valves grow with the child).
Cardiac patch technology — small grafts of engineered heart tissue applied to damaged areas of the heart following a myocardial infarction — has reached human trials. The Charité-Universitätsmedizin Berlin patches and the BioVAT-HF programme in Germany are both currently in trials with promising early data. The patches are not whole-organ replacement, but they’re a meaningful intervention for patients who would otherwise progress to heart failure.
Stem Cell Sources: From iPSC to Direct Reprogramming
One area where the documentary’s account has been substantially refined is on the cell-source question. The original framing assumed that pluripotent stem cells would come either from embryonic sources (with attendant ethical and regulatory challenges) or from adult bone-marrow-derived stem cells (which turned out to be less plastic than initially hoped). The breakthrough that re-shaped the field — Shinya Yamanaka’s induced pluripotent stem cell (iPSC) work, which earned the 2012 Nobel — was new enough at broadcast time to be touched on but not central.
The picture now: iPSCs derived from a patient’s own skin or blood cells, reprogrammed back to pluripotency and then differentiated into cardiomyocytes, are the dominant cell source for heart tissue engineering work. Patient-specific iPSCs eliminate the immune-rejection problem entirely (the cells are the patient’s own genome) and avoid the ethical questions around embryonic sources. The remaining challenge is differentiation efficiency and consistency — producing reliably uniform batches of mature cardiomyocytes at scale.
An adjacent technique that has emerged since the documentary aired is direct reprogramming — converting one differentiated cell type directly into another without the intermediate pluripotent step. Proof-of-concept work by Deepak Srivastava’s lab and others has demonstrated direct conversion of cardiac fibroblasts (which are abundant in scar tissue after a heart attack) into beating cardiomyocytes within the heart itself, using a cocktail of three or four transcription factors. If this approach matures, it could allow regeneration of damaged heart tissue without external scaffold or transplantation at all.
The Clinical Reality, Today
Where the technology actually stands in the clinic, as of the most recent published data: bioengineered heart valves are in routine paediatric use. Cardiac patches are in human trials with regulatory approval expected within the next several years for specific indications. Whole-heart bioengineered transplants remain pre-clinical — no human has yet received an engineered whole heart and the leading research groups generally suggest this is at least a decade away.
For the foreseeable future, the dominant clinical pathway for end-stage heart failure remains either donor-organ transplantation (limited by the well-known shortage of donors) or mechanical circulatory support (left ventricular assist devices, which have improved dramatically and are now used both as bridges to transplant and as long-term destination therapy). Tissue engineering is the long-term hope, not the near-term answer.
The Wider Tissue-Engineering Landscape
The documentary touched briefly on tissue engineering work outside the heart — skin, bladders, tracheas, ears — and the field has continued to develop unevenly. Some areas have moved into clinical practice (engineered skin grafts for burn patients are routine; engineered bladders for specific congenital conditions have been used in small numbers of patients with reasonable outcomes). Others have struggled (the engineered trachea work led by Paolo Macchiarini ended in scandal after several patient deaths and the retraction of multiple papers; the field has spent the years since rebuilding scientific rigour and ethical oversight).
The general lesson Hugo drew from the rewatch is that tissue engineering is a discipline that genuinely is going to transform medicine — but on a timescale measured in decades rather than years, and with the kind of slow, unglamorous, peer-reviewed grind that doesn’t always make for compelling television. Documentaries that promise imminent breakthroughs in any biomedical field should be viewed with suspicion. The honest version of this science is patient.
Where the Documentary Got It Right
Three things our team consistently flagged. First, the lab access — the production secured genuine working access to several of the key research labs (Doris Taylor’s, Anthony Atala’s at Wake Forest, the Mass General Hospital cardiac group) and showed actual research as it happens. Real biomedical research mostly looks like microscopes and sterile-technique pipetting, which is less photogenic than the public expects. The documentary handled this well, treating the work with the weight it deserves rather than over-dramatising.
Second, the patient interviews. The documentary included multiple interviews with patients on heart-transplant waitlists, in artificial-heart trials, or living with severe heart failure. Their accounts grounded the abstract science in why it matters. That editorial choice was the right one.
Third, the appropriate hedging. The narration consistently framed the work as research toward a future therapy rather than an imminent clinical option. Most science television fails this test; this one passed.
Where We’d Push Back
The pacing of the timeline. Several segments implied that whole-organ engineering was within five to ten years of clinical use. We’re now thirteen years past broadcast and that timeline has slipped substantially. We don’t blame the producers — the field’s leading researchers were similarly optimistic at the time — but the lesson for any current science television is to flag the gap between proof-of-concept and clinical reality more aggressively. Most “breakthrough” research stalls in the long, expensive translation to humans.
What to Watch and Read Alongside
For follow-up reading, our team’s recommendations: Hannah Devlin’s The Genetic Lottery for context on stem-cell biology; the review papers by Anthony Atala on tissue engineering at Wake Forest (open-access, written for a clinical audience but accessible to determined non-specialists); and for the broader history of organ replacement, David Hamilton’s A History of Organ Transplantation.
For viewing, the recent NOVA episode 3D Printing the Body (2018) covers the bioprinting side of tissue engineering, which has matured considerably since the original documentary. The BBC’s Frontiers radio series has done several solid episodes on the iPSC story.
The bigger picture, in Hugo’s words from his lab days: tissue engineering is not a single discovery away from changing medicine. It is a long arc of incremental advances in cell biology, materials science, and clinical practice that together will, eventually, give us the ability to replace failed organs with engineered ones. We’re somewhere in the middle of that arc. The documentary captured an important early stage of it. The next stage is now.




